- Name
- Barbara Benham
- bbenham1@jhu.edu
- Name
- Robin Scullin
- rsculli@jhu.edu
A genetic defect tied to a variety of neurodegenerative diseases and mental illnesses changes how cells starved of sugar metabolize fatty compounds known as lipids, a new study led by researchers from the Johns Hopkins Bloomberg School of Public Health shows. The finding could lead to new targets to treat these diseases, which currently have no cure or fully effective treatments.
Researchers identified the mutation in the C9orf72 gene several years ago, and it has since been associated with amyotrophic lateral sclerosis (also known as ALS), frontotemporal dementia, Alzheimer's disease, bipolar disorder, and other neurologic diseases. Although researchers have known that this mutation decreases the amount of the protein coded by the C9orf72 gene in cells, it was unknown exactly what this C9orf72-coded protein does.
The findings, published online in the journal Genes & Development, show that the genetic defect leads to greater amounts of a protein that causes cells to overproduce lipids and an enzyme called NOX2, which is known to cause oxidative stress that can damage cells.
"Cells with this mutation act as if they're chronically under stress, which could underlie the pathology of diseases associated with this defect," says Jiou Wang, an associate professor in the Bloomberg School's Department of Biochemistry and Molecular Biology. "Our study raises the question of whether we should be looking at problems with lipid metabolism as a potential cause for these diseases."
To learn its function, the researchers created mouse cells in which C9orf72 was effectively removed, preventing the production of its protein in order to study what happens in cells naturally carrying the gene's mutated form. They compared these cells to those with the gene, looking broadly at the levels of all proteins produced in both types of cells under normal conditions. Previous studies have hinted that C9orf72 might play a role in how cells protect themselves from nutrient starvation stress, Wang explains, so researchers also compared protein levels when cells were exposed to environments devoid of glucose, an important cellular energy source.
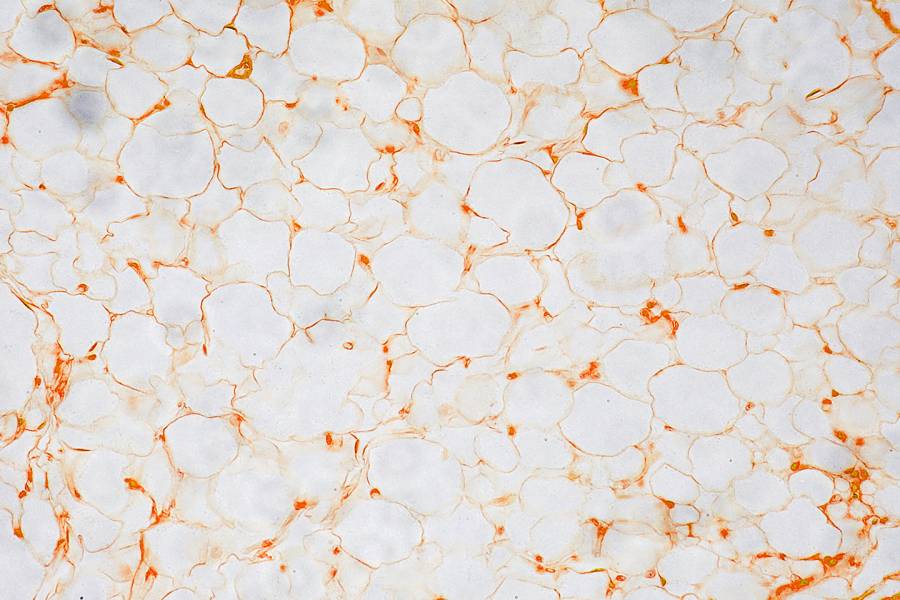
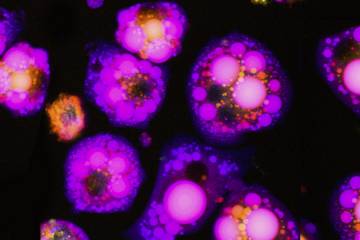
When healthy cells are deprived of glucose, Wang says, they tend to make and store more lipid droplets. However, the researchers' protein level analysis showed that when deprived of glucose, the cells lacking the C9orf72 protein produced significantly more lipid metabolism-related proteins compared to cells with this gene. When the researchers compared the number of lipid droplets between the two types of cells in glucose-devoid conditions, the cells lacking C9orf72 protein held significantly more. They also had more free fatty acids, the individual components that come together to form these lipid droplets.
The researchers found that in cells without the C9orf72 gene, both cellular pathways used to create lipid droplets—de novo lipid biogenesis, when the cell makes lipid droplets from scratch, and autophagy, when cells digest other cell components to create lipids—were abnormally regulated. They determined which other proteins are associated with C9orf72 and found that CARM1, a protein known to broadly affect which genes produce proteins and how much, was affected. In cells without C9orf72, the CARM1 protein levels increased, leading to greater expression of genes related to lipid production.
Taken together, Wang says, these results suggest that mutations in C9orf72 lead to greater amounts of CARM1, which causes cells to overproduce lipids and NOX2 in response to glucose starvation.
"As we learn more about this newly discovered biological pathway," says Wang, "it could lead to new therapeutic interventions that protect cells that carry this mutation from harm."
Posted in Health, Science+Technology
Tagged alzheimer's disease, dementia, als, microbiology, bipolar disorder