- Name
- Vanessa Wasta
- wasta@jhmi.edu
- Office phone
- 410-955-8236
Researchers from the Johns Hopkins University School of Medicine have helped develop a nanobody capable of getting through the tough exterior of brain cells and untangling misshapen proteins that lead to Parkinson's disease, Lewy body dementia, and other neurocognitive disorders.
The research, published last month in Nature Communications, was led by Xiaobo Mao, an associate professor of neurology at the School of Medicine, and included scientists at the University of Michigan, Ann Arbor. Their aim was to find a new type of treatment that could specifically target the misshapen proteins, called alpha-synuclein, which tend to clump together and gum up the inner workings of brain cells. Emerging evidence has shown that the alpha-synuclein clumps can spread from the gut or nose to the brain, driving the disease progression.
Nanobodies—miniature versions of antibodies, which are proteins in the blood that help the immune system find and attack foreign pathogens—are natural compounds in the blood of animals such as llamas and sharks and are being studied to treat autoimmune diseases and cancer in humans. In theory, antibodies have the potential to zero in on clumping alpha-synuclein proteins, but have a hard time getting through the outer covering of brain cells. To squeeze through these tough brain cell coatings, the researchers decided to use nanobodies instead.
The researchers had to shore up the nanobodies to help them keep stable within a brain cell. To do this, they genetically engineered them to rid them of chemical bonds that typically degrade inside a cell. Tests showed that without the bonds, the nanobody remained stable and was still able to bind to misshapen alpha-synuclein.
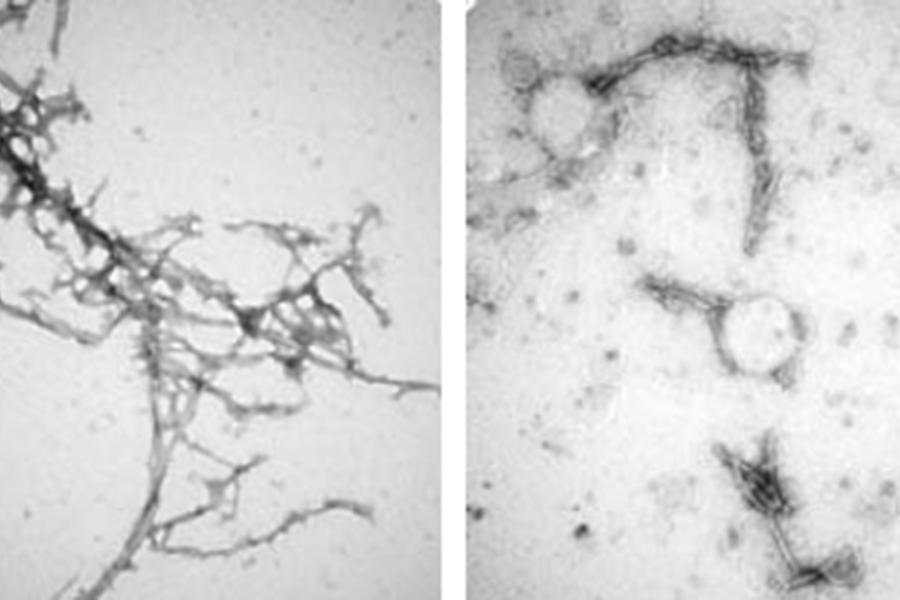
The team made seven similar types of nanobodies, known as PFFNBs, that could bind to alpha-synuclein clumps. Of the nanobodies they created, one—PFFNB2—did the best job of glomming onto alpha-synuclein clumps and not single molecules, or monomer of alpha-synuclein, which are not harmful and may have important functions in brain cells.
Additional tests in mice showed that the PFFNB2 nanobody cannot prevent alpha-synuclein from collecting into clumps, but it can disrupt and destabilize the structure of existing clumps.
"Strikingly, we induced PFFNB2 expression in the cortex, and it prevented alpha-synuclein clumps from spreading to the mouse brain's cortex, the region responsible for cognition, movement, personality, and other high-order processes," says Ramhari Kumbhar, the co-first author and a postdoctoral fellow at the School of Medicine.
"The success of PFFNB2 in binding harmful alpha-synuclein clumps in increasingly complex environments indicates that the nanobody could be key to helping scientists study these diseases and eventually develop new treatments," Mao says.
Other researchers involved in this study include Yuqing Liu, Ramhari Kumbhar, Kundlik Gadhave, and Ning Wang of the Johns Hopkins University School of Medicine; Yemima R Butler, Wenjing Wang, and Peiran Zhao of the University of Michigan, Ann Arbor; and Yanmei Li of Tsinghua University.
Posted in Health, Science+Technology
Tagged neuroscience, parkinson's disease, nanomaterials