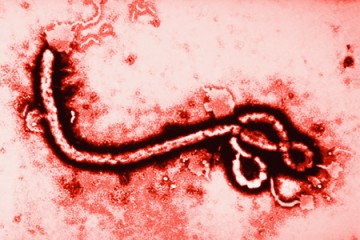
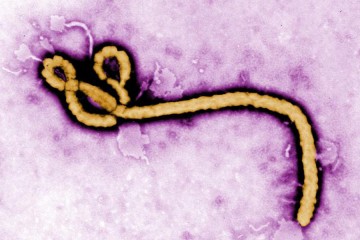
Johns Hopkins bioethicist Nancy Kass defends the unique treatment given to the two Americans infected with Ebola but advises proceeding with caution for wider distribution of the experimental drug in an opinion piece published Monday in the Annals of Internal Medicine.
Two American aid workers who contracted the Ebola virus in Africa have been treated at Emory University Hospital in Atlanta with a drug called ZMapp, a cocktail of plant-derived antibodies that has never before been used to treat humans. Because the drug was still being tested at the time of the latest Ebola outbreak, it is in very short supply.
Kass, the Deputy Director for Public Health at the Johns Hopkins Berman Institute of Bioethics, addresses a question on many minds: If it was reasonable to treat these two Americans, shouldn't the experimental treatment also be provided to African patients?
"Callous as it may seem, the answer is no, or at least, not yet," she writes. "The threshold for determining that an individual patient receive access to a highly experimental drug on a compassionate use basis is, and ought to be, different from the threshold for rolling out a treatment program to an entire community, even one facing a life threatening epidemic."
Watch: Medical inequality in Ebola treatment? (MSNBC)
Kass cautions that a rapid, widespread rollout could backfire, to the long-term detriment of public health.
"In addition to considering safety risks of untested treatments, the risk they will be ineffective also must be weighed," she writes. "A top priority in this, and any, public health emergency is public health cooperation—those affected must be willing to follow directions from international and local health authorities. ... What is at stake in a decision about using experimental treatments may be far greater than the risks to the individuals who take them."
Kass says a gradual approach to treatment is prudent and that "any roll out must proceed with caution."
"Starting with small numbers is itself a protection, as is unwavering commitment to ongoing data collection," she notes. "Ultimately, such data will be our best ethical guide."
Posted in Health, Voices+Opinion
Tagged public health, bioethics, berman institute of bioethics, ebola