Inspired by how human bone and colorful coral reefs adjust mineral deposits in response to their surrounding environments, Johns Hopkins researchers have created a self-adapting material that can change its stiffness in response to the applied force. This advancement can someday open the doors for materials that can self-reinforce to prepare for increased force or stop further damage.
A report of the findings was published in March in Advanced Materials.
"Imagine a bone implant or a bridge that can self-reinforce where a high force is applied without inspection and maintenance. It will allow safer implants and bridges with minimal complication, cost, and downtime," says Sung Hoon Kang, an assistant professor in the Department of Mechanical Engineering, Hopkins Extreme Materials Institute, and Institute for NanoBioTechnology and the study's senior author.
While other researchers have attempted to create similar synthetic materials before, doing so has been challenging because such materials are difficult and expensive to create, or require active maintenance when they are created and are limited in how much stress they can bear. Having materials with adaptable properties, like those of wood and bone, can provide safer structures, save money and resources, and reduce harmful environmental impact.
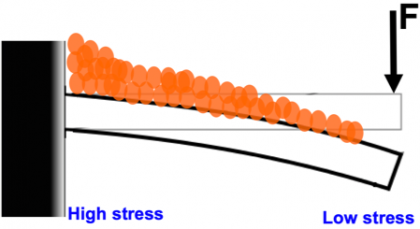
Image caption: More stress placed on one end of the beam led to more mineralization. As stress gradually decreased across the beam, so did the amount of mineralization.
Image credit: Sung Hoon Kang
Natural materials can self-regulate by using resources in the surrounding environment; for example, bones use cell signals to control the addition or removal of minerals taken from blood around them. Inspired by these natural materials, Kang and colleagues sought to create a materials system that could add minerals in response to applied stress.
The team started off by using materials that can convert mechanical forces into electrical charges as scaffolds, or support structures, that can create charges proportional to external force placed on it. The team's hope was that these charges could serve as signals for the materials to start mineralization from mineral ions in the environment.
Kang and colleagues immersed polymer films of these materials in a simulated body fluid mimicking ionic concentrations of human blood plasma. After the materials incubated in the simulated body fluid, minerals started forming on the surfaces. The team also discovered that they could control the types of minerals formed by controlling the fluid's ion composition.
The team then set up a beam anchored on one end to gradually increase stress from one end of the materials to the other and found that regions with more stress had more mineral buildup; the mineral height was proportional to the square root of stress applied.
Their methods, the researchers say, are simple, low-cost and don't require extra energy.
"Our findings can pave the way for a new class of self-regenerating materials that can self-reinforce damaged areas," Kang says. He hopes that these materials can someday be used as scaffolds to accelerate treatment of bone-related disease or fracture, smart resins for dental treatments or other similar applications.
Additionally, these findings contribute to scientists' understanding of dynamic materials and how mineralization works, which could shed light on ideal environments needed for bone regeneration.
Posted in Science+Technology
Tagged biomedical research









