- Name
- Rachel Butch
- rbutch1@jhmi.edu
- Office phone
- 410-955-8665
- Name
- Vanessa Wasta
- wasta@jhmi.edu
- Office phone
- 410-955-8236
Johns Hopkins researchers report they have demonstrated in mouse tissue grown in the lab that the cell layer surrounding breast milk ducts reaches out to grab stray cancer cells to keep them from spreading through the body. The findings reveal that this cell layer, called the myoepithelium, is not a stationary barrier to cancer invasion, as scientists previously thought, but an active defense against breast cancer metastasis.
Results of the scientists' experiments were published online this week in the Journal of Cell Biology.
"Understanding how cancer cells are contained could eventually help us develop ways to predict a person's individualized risk of metastasis," says Andrew Ewald, a professor of cell biology at the Johns Hopkins University School of Medicine and a member of the Johns Hopkins Sidney Kimmel Comprehensive Cancer Center.
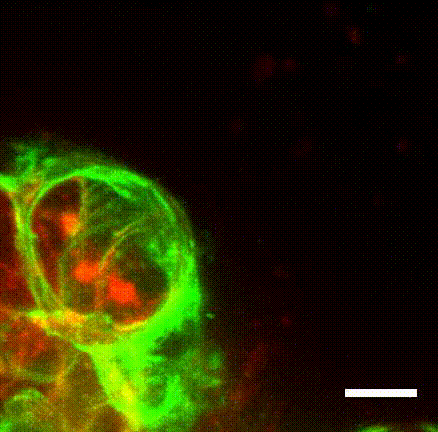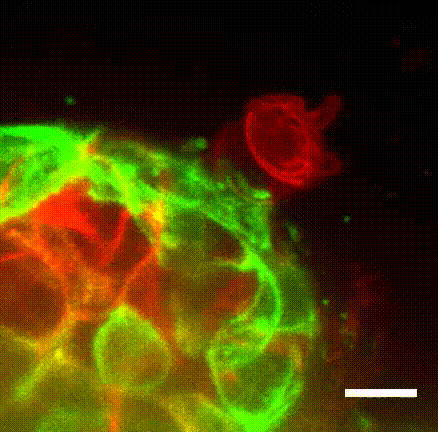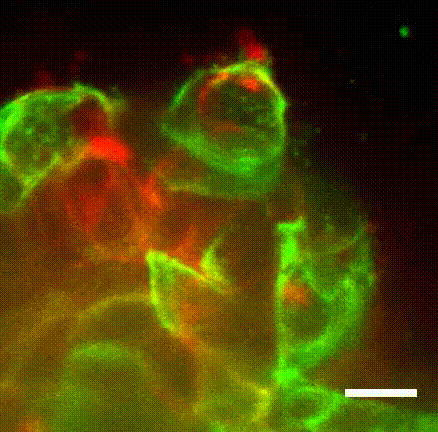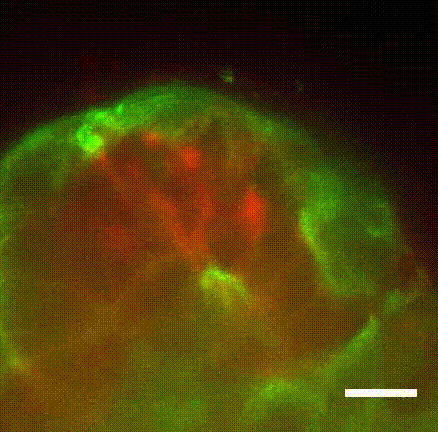
Image caption: The myoepithelium (green) dynamically restrains a protruding Twist1+ luminal cell (red), which has been linked to cancer metastasis in multiple tumor types.
Image credit: Ewald Laboratory
Most breast tumors begin in the cells that line the interior of breast milk ducts. These cells in turn are surrounded by myoepithelial cells, Ewald says, which work together to contract and move milk through the ducts when a baby is nursing.
This myoepithelial layer is used clinically to distinguish contained breast cancers from invasive cancers in humans. When breast cancer cells breach the myoepithelial layer, the result is so-called invasive carcinoma, which is associated with higher rates of recurrence and the need for more aggressive treatment, says Ewald.
"If you think about metastasis as a long race, breaking through this layer is the exit from the starting gate," Ewald says.
For their study, Ewald and his team engineered cells taken from the lining of mouse breast ducts to produce the protein Twist1, which works by altering gene expression and which has been linked to cancer metastasis in multiple tumor types.
To their surprise, the researchers saw that when the invasive Twist1 cells broke through the myoepithelial layer, the myoepithelial cells grabbed the cells that had gone astray and successfully pulled them back within the breast duct lining 92 percent of the time through 114 observations.
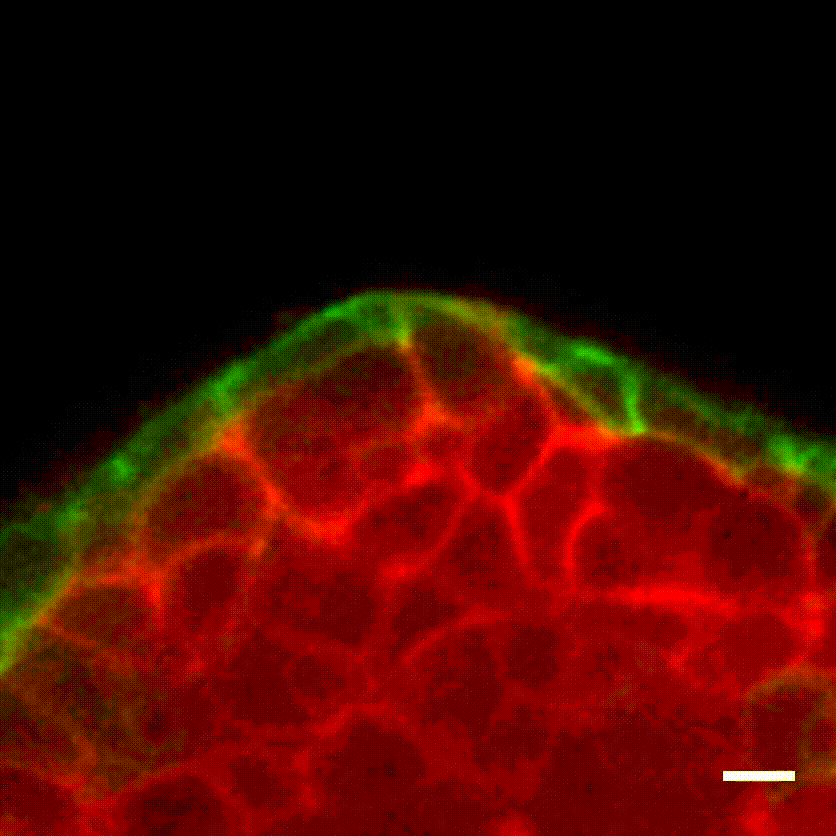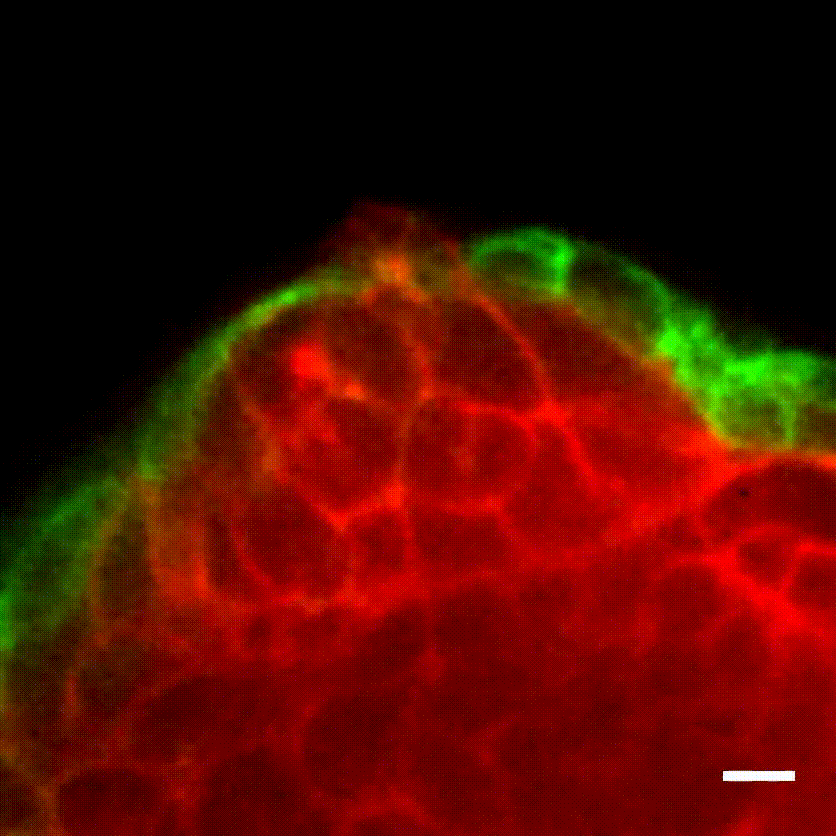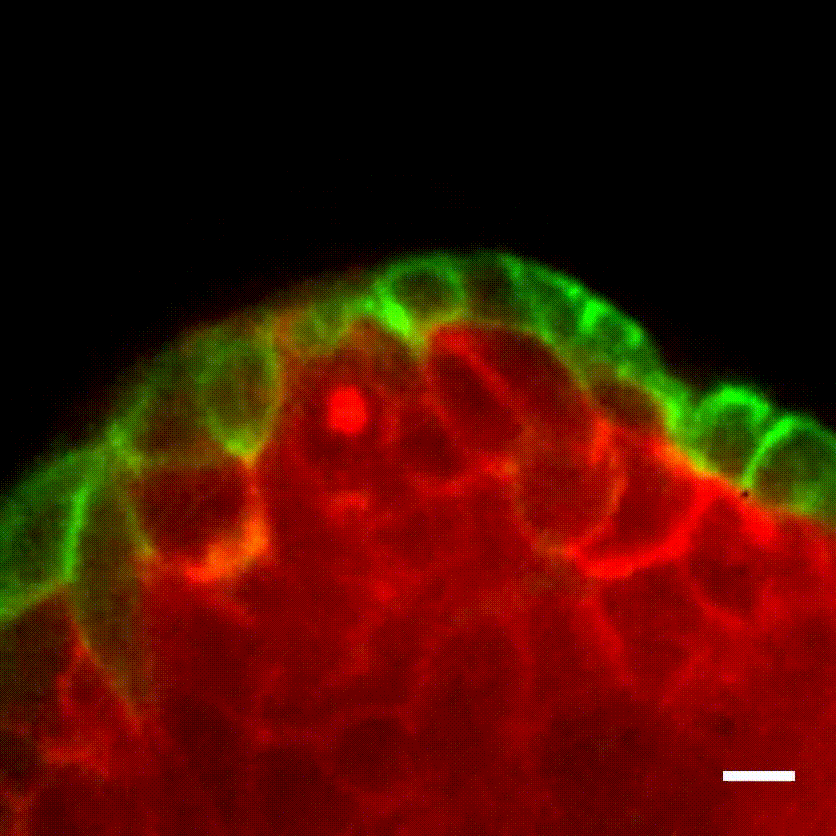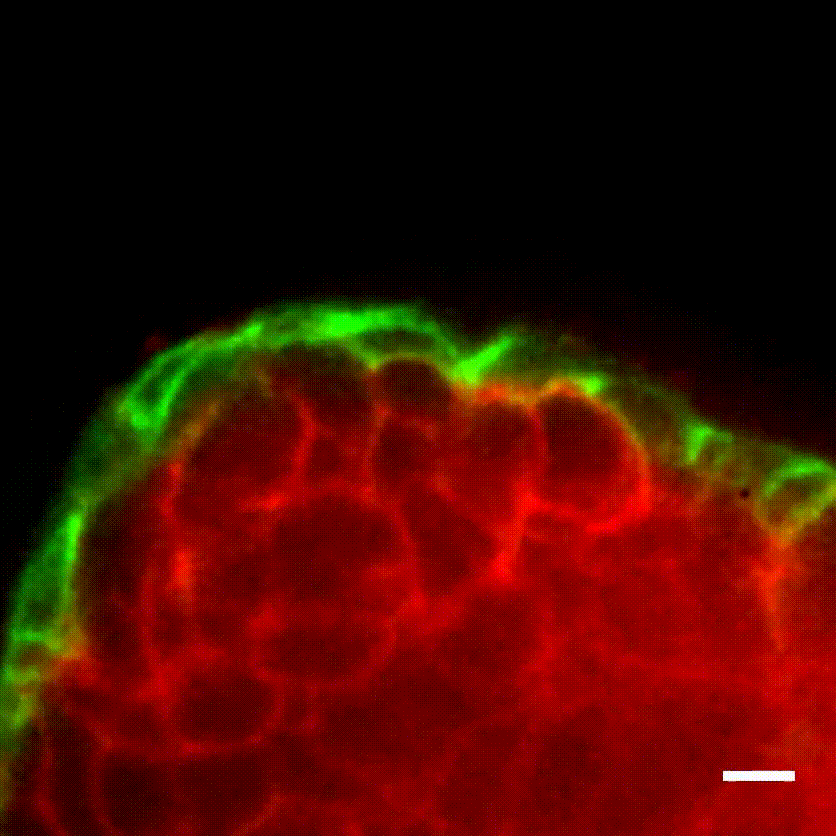
Image caption: The myoepithelium (green) restrains from escape a protruding Twist1+ luminal cell (red).
Image credit: Ewald Laboratory
"These findings establish the novel concept of the myoepithelium as a dynamic barrier to cell escape, rather than acting as a stone wall as it was speculated before" says Katarina Sirka, a PhD student in Ewald's laboratory.
To confirm that their findings indicated active behaviors, Ewald and his team altered two key characteristics of myoepithelial cells—their ability to contract and their numerical ratio to the invasive cells.
First, the researchers genetically engineered mouse myoepithelial cells to deplete their smooth muscle actin, a protein that allows the cells to contract. Under that condition, the number of escaped invasive cells that broke through the myoepithelial layer increased threefold compared to control cells with a normal myoepithelium.
Likewise, the researchers found that decreasing the proportion of myoepithelial cells to invasive cells increased the number of escaped cancer cells. By adding just two myoepithelial cells for each invasive cell, the escape rate decreased fourfold compared with the spread of invasive cells with no defending barrier.
"This is important to know because it suggests that both the physical completeness of the myoepithelium and the gene expression within the myoepithelial cells are important in predicting the behavior of human breast tumors. Anywhere this layer thins or buckles is an opportunity for cancer cells to escape," says Eliah Shamir, a surgical pathology fellow at the University of California, San Francisco, who contributed to the study.
In the future, Ewald and his team plan to study the cellular mechanisms prompting the myoepithelial layer to react so dynamically and what makes it fail during invasive progression.
Posted in Health
Tagged cancer, breast cancer, kimmel cancer center, cancer metastasis