Researchers at Johns Hopkins University say they have discovered how a common gene mutation causes the brain damage associated with dementia and amyotrophic lateral sclerosis—better known as ALS, or Lou Gehrig's disease—and are experimenting with a molecular therapy to address the problem.
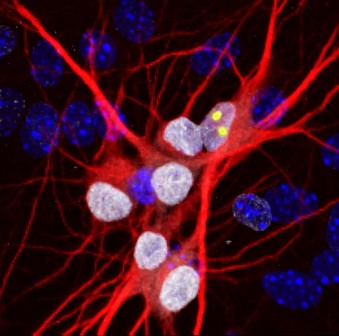
Image caption: Neurons (red) created from ALS patients bearing the C9orf72 mutatio show clumps of the RanGAP protein (yellow) in their nuclei (white). The nuclei of other cells are in blue.
Image credit: Jeffrey Rothstein laboratory, Johns Hopkins Medicine
The research shows that the most common known risk factor for ALS and frontotemporal dementia, or FTD—a gene mutation known as C9orf72—causes cellular highways to jam, impairing brain functions. However, in tests with human and fly cells, the scientists were successful in using a molecular therapy to clear up those jams.
A report on this research is published in the Sept. 3 issue of the journal Nature .
Both ALS and FTD are characterized by degeneration of nerve cells over time. In FTD, the damage causes problems with speech, understanding language, and processing emotions. In ALS, the degeneration affects cells in the spinal cord as well as the brain, and patients gradually lose muscle control.
Several years ago, the discovery of the gene mutation linked to ALS and FTD "was really a game changer for the field because it wasn't a typical genetic mutation," says Jeffrey Rothstein, a professor of neurology and director of the Brain Science Institute and the Robert Packard Center for ALS Research at the Johns Hopkins School of Medicine. "Now we have some information about what it is doing early on to damage brain and spinal cord cells."
Scientists have seen that the C9orf72 mutation associated with the two diseases causes long strands of RNA to clog pathways that normally move proteins into a cell's nucleus.
To mediate the effect of the mutated RNA, the research group has focused on one protein in particular, RanGAP. Researchers identified this protein as a critical target of C9orf72 repeats that could prevent brain cell death when its function is restored, according to Thomas Lloyd, an associate professor of neurology at Johns Hopkins, whose team worked with Rothstein's on this research.
In one set of experiments with fly and human stem cells, the scientists found a way to block the repetitive RNA strands from interacting with the RanGAP protein. As a result, the jammed up nuclear pores reopened, allowing key proteins to move once again.
It's possible this effect could be replicated in a drug for patients with ALS and FTD, said Rothstein, who has launched a collaboration with California-based Isis Pharmaceuticals. But he cautioned that further studies are necessary before any such commercially available drug is developed.
"We still don't know every step between the C9orf72 mutation and cellular death in the brain," Rothstein says. "But our belief is that this is what starts it off, and this is certainly a good therapeutic target."
Read more from Hopkins MedicinePosted in Health, Science+Technology








