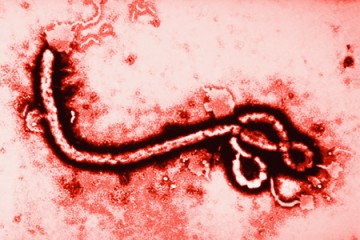
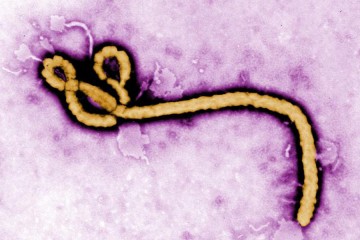
In August of 2014, a small biotech company in San Diego with just nine employees became international news. Mapp Biopharmaceutical appeared to have a cure for Ebola. Called ZMapp, the experimental treatment was given to two American aid workers infected with the deadly virus. The patients survived, and a media frenzy ensued. Larry Zeitlin, Mapp's president, and Kevin Whaley, chief executive officer, were inundated with press requests. Everyone from the BBC to Bloomberg News, from CBS to Fox, ran stories on the new treatment. Zeitlin told The New York Times that the sudden demand for ZMapp, which was limited in quantity and had never been tested in humans before being given to the patients, was "absolutely overwhelming."
Zeitlin, 46, and Whaley, 63, are at the helm of one of the few biopharmaceutical companies to successfully manufacture human monoclonal antibodies in plants. The human immune system naturally develops antibodies to fight toxins. Some pathogens, however, overwhelm the body's defense system and we cannot produce antibodies fast enough. Decades ago, scientists discovered that when they took blood from a person who has survived a disease, extracted the right antibodies, and then put those into someone else, the recipient would become passively immune to that infection. An example of passive immunity is the shot you get for malaria before traveling.
Antibodies have traditionally been manufactured in mammalian cell culture, which is expensive. Zeitlin and Whaley, along with research partners, figured out how to manufacture the three antibodies for ZMapp using tobacco plants. Plantibodies, as these are called, are not yet commercially available. Today, there's just one FDA-approved drug made by using a plant. Called Elelyso, the drug is an enzyme manufactured using carrot cells and administered to treat the hereditary condition Gaucher disease. "[Producing antibodies in plants] is not something that is fully embraced by the pharmaceutical industry, and with good reason," Whaley says. "In a lot of ways, it's still an unproven technology."
This is research that the two first began in the lab of Johns Hopkins biophysicist Richard Cone. Whaley and Zeitlin met there in the 1980s and worked with Cone and Thomas Moench, a physician specializing in infectious disease, on developing antibodies as a contraceptive as well as protection against STDs. "Richard's lab was most interested in these big public health problems," Zeitlin says. "I loved that [the researchers in Cone's lab] were applying science to not just understand how things work, but to solve problems, to try to make a contraceptive, to try and solve STDs. I was hooked."
He and Whaley became interested in plantibodies in the late 1980s. The promise of plants, Whaley explains, is the ability to manufacture treatments for less money. "You can't go to Africa and charge people $1,000 a year for contraception. You need a way to make things cheaper," Zeitlin says. He and Whaley left Johns Hopkins in 1999 to work for a California startup that made human monoclonal antibodies in corn plants, but the company didn't survive. "They filed for bankruptcy and we were both out of jobs," Zeitlin says. It was Whaley's idea to start their own company. "I was quite nervous about it, but it seems be working out," Zeitlin says.
The two had learned a few things at that failed startup. "The company was making antibodies in corn," Zeitlin says, "then people started to realize that making pharmaceuticals in food crops isn't a good idea." The other problem with corn: It takes too long to grow. Testing antibodies against pathogens is an iterative process that requires some guesswork. When the human body fights a disease, it throws a host of antibodies at the problem. The trick for scientists, then, is to pick which of the antibodies are the most effective against any given disease. This requires trial and error, picking one antibody, manufacturing it in the lab, testing it against the disease. The researchers needed to produce new antibodies for testing at a more rapid pace. "Where it took a year and half in corn, it took just weeks with tobacco," Zeitlin says.
With funding from Department of Defense and NIH grants, Mapp focused on antibody research for treating deadly viruses and poisons, as well as continuing research in partnership with Cone for sexually transmitted diseases. By 2010, the Defense Advanced Research Projects Agency created a project called Blue Angel. Plantibodies could mean cheaper, faster ways to develop treatment against virulent epidemics or man-made bioterrorism. "They decided that plants could be a transformative technology," Zeitlin says. DARPA funded four new facilities to manufacture antibodies in plants, including Kentucky BioProcessing, where ZMapp is made.
While the media is focused on ZMapp, Whaley says the real potential of the drug isn't just its efficacy in treating Ebola—it's what that treatment and their research portends for a host of seemingly intractable illnesses and conditions, particularly for people living in poverty. At Mapp, Whaley and Zeitlin not only seek treatments for rare and dangerous viruses and poisons—Ricin, Ebola, Marburg, Junin—they have also advanced the use of human monoclonal antibodies for contraception and protection against sexually transmitted diseases like HIV. "ZMapp is not really the story. The real story is about antibodies and how diverse they are. ZMapp shows the power of antibody-based technology, and that's the exciting thing to us. They work; they are effective," Whaley says. "So now can we produce antibodies at a low enough cost and at a large enough capacity so that it plays a role in global health?" That's what Zeitlin and Whaley hope to find out next.
Posted in Health
Tagged ebola, richard cone, experimental drugs, zmapp, cone lab, mapp biopharmaceutical