Not long ago, here is what Andrew Ewald did to study cancer. Instead of starting with the conventional research cell lines, he got breast cancer cells from the primary tumors of actual patients. That is, not models standing in for cancer—the real thing. And instead of putting those malignant cells into the artificial two-dimensional environment of standard, planar petri dishes, he embedded them in three-dimensional gels of his own design that mimic the matrix of collagen fibers in the human body. Then he rigged microscopes to photograph not just a single tiny area but hundreds of different positions within a thick swath of cells and gel. He took thousands of pictures, one every 10 to 20 minutes, day after day, photographing hundreds of samples at a time. It was a long, arduous process, but Ewald, a professor of cell biology at the School of Medicine and a member of the Kimmel Cancer Center, had his reasons.
Cancer is a complex disease—actually a complex collection of complex diseases. But in the laboratory, researchers have typically studied it by isolating lab-produced malignant cells in the simplest of environments. Some years ago, skeptical scientists began to question these methods, observing, for example, that cancer cells in a petri dish behave so differently from tumors in a human body as to cast doubt on much conventional research. Ewald is at the vanguard of a generation of scientists determined to deal with cancer's full complexity.
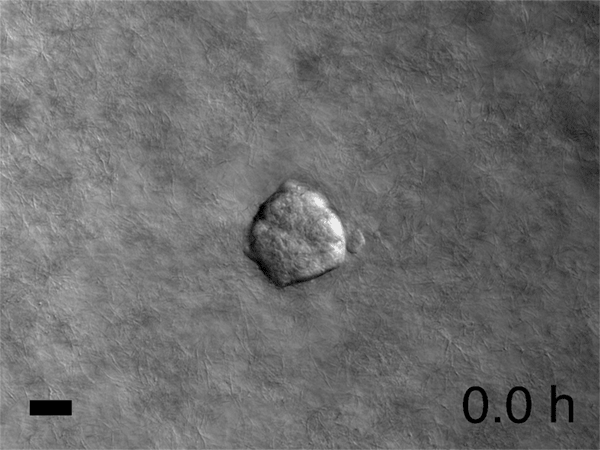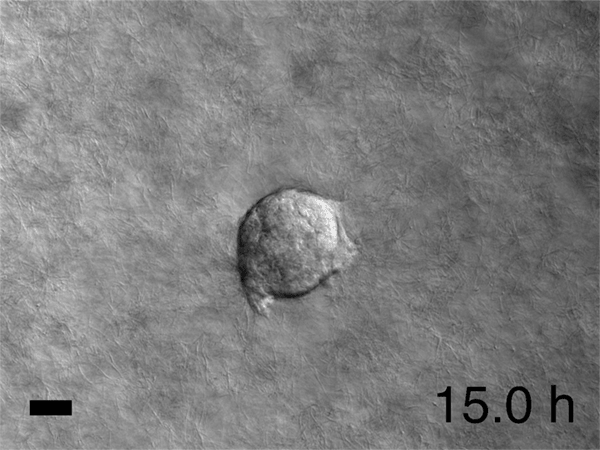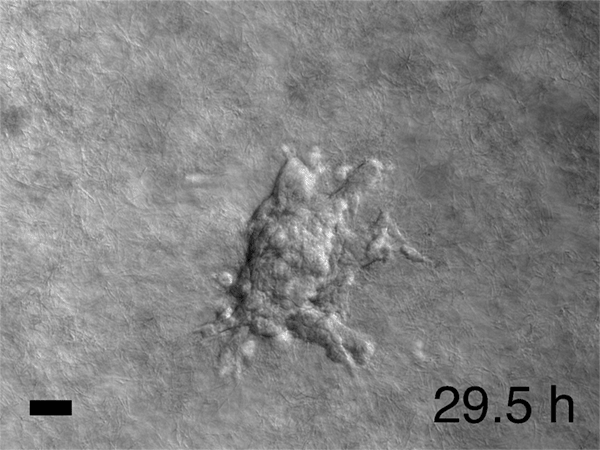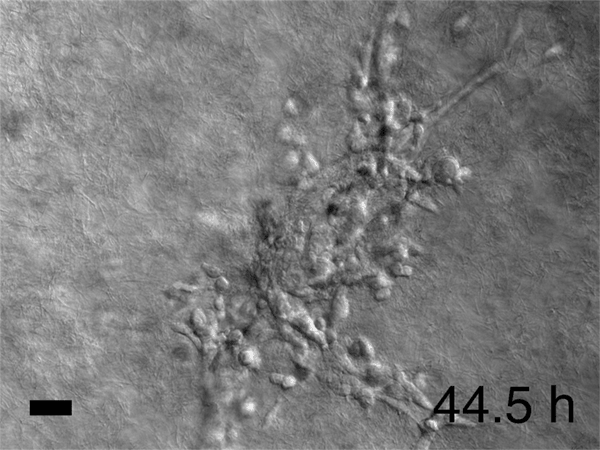
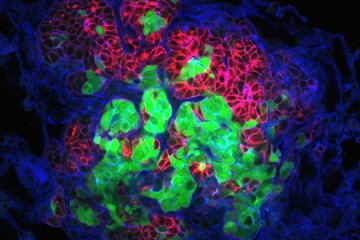
Image caption: Breast cancer tumor cells invade a 3D environment composed of collagen I. This model could support the discovery of new drugs to stop cancer metastasis.
Image credit: Courtesy of Andrew Ewald
In his lab, he and his colleagues watched time-lapse movies, assembled from those thousands of microphotographs, of what happened to the cancer cells they'd embedded in gel. Startled, they saw slices of a tumor grow what looked like tiny tentacles. The tentacles were cells migrating out from the tumor into the gel along fibrous tracks that resembled cellular highways. Each invading group clearly had leader cells at the front of the pack, pulling followers behind them. Scientists had observed similar activity before, but no one had expected to see that each pack was led by a specific cell that seemed specialized for the job. Was this just a quirk of the tumor Ewald and his colleagues happened to be studying? They repeated the experiment using cells from the other two major breast cancer variants. Each showed the same cell type at the head of the march. "It was literally a hair-standing-on-the-back-of-the-neck, shivers-down-the-spine moment," Ewald says. Had cancer just revealed one of its fundamental deadly tricks?

Ewald is a cancer biologist who has never taken a formal university course in cancer biology. He trained as a physicist and comes at cancer as, essentially, a really interesting and hard physics problem. What are the fundamental factors that make cancer behave the way it does? What is the E = mc2 of cancer? "I'm not very good at math anymore," he says. "But two things have stuck with me from physics. The first is a facility with instrumentation"—his ability to customize microscopes, for example—"and the second is a hatred of uncontrolled variables."
When the physics-trained Ewald began attending cancer conferences in the early 2000s as a postdoctoral researcher at the University of California, San Francisco, he realized how differently biologists approached the disease compared to what a physicist would do. Cancer is mind-bogglingly complicated. Every tumor contains groups of cells with different genetic mutations, and even within genetically identical groups, genes are expressed at different levels. Those cells interact with the body's connective fibers, blood vessels, immune cells, and so on. They're subject to varied forces and pressures. And that's just one tumor. Each kind of breast cancer tumor differs substantially from the others, and even more from tumors produced by lung cancer, brain cancer, pancreatic cancer, and so on.
Ewald knew of many studies that showed how changing a gene's expression or a single environmental variable could affect how a particular tumor grew. But what, he wondered, about the nine or 10 other factors that also influenced the tumor in unknown ways that the experimentalist could not control? A physicist studying something might use vacuum chambers, cryogenic temperatures, magnetic shielding, and other tricks to try to quiet confounding factors and ensure that any observed effects were from the one variable under scrutiny. Biologists had no comparable methods for filtering the complications of cancer.
"There's a belief in underlying simplicity that has guided physics since forever," Ewald says. "Where we see complication, that's where we don't understand the principles yet. We don't understand the underlying simplicity." Think about an empty room. The room might contain, say, 1025 air molecules. To describe the physics of the air, you might try to write down 1025 kinematic equations, each one describing the motion of a single molecule. But if instead you measured the air's temperature and put that value into a single equation developed by 19th-century physicists James Clerk Maxwell and Ludwig Boltzmann, you would know everything you need to know about the speed of the molecules. That's what physics does—extracts fundamental simplicity from a seemingly hopeless mess of complexity.
That quest for simplicity, however, comes with an important caveat. You may have heard this aphorism, often attributed to Albert Einstein: "Everything should be made as simple as possible, but not simpler." (Einstein actually said, "It can scarcely be denied that the supreme goal of all theory is to make the irreducible basic elements as simple and as few as possible without having to surrender the adequate representation of a single datum of experience." Less pithy, for sure, but the same idea.) Much current research aligns with the idea that cancer is primarily a matter of faulty genes. It's an attractive view because the key to preventing and curing the disease then becomes identifying and fixing or suppressing bad genes; other complicated aspects of cancer biology—a tumor's physical environment, how it interacts with other kinds of cells, the mechanics of how it invades the rest of the body—become secondary. When Bill Clinton announced the completion of the Human Genome Project in 2000, he said, "In coming years, doctors increasingly will be able to cure diseases like Alzheimer's, Parkinson's, diabetes, and cancer by attacking their genetic roots. ... In fact, it is now conceivable that our children's children will know the term 'cancer' only as a constellation of stars." Seventeen years later, it's clear that the gene-centric view of cancer is not the full picture, and while it is pointing the way to some useful therapies, it cannot single-handedly cure the disease. Perhaps that's because, in its simplicity, it leaves out crucial information—Einstein's data of experience.
For many decades, physicists have built tools important for cancer research and therapy. Microscopy, for example, is the harnessing of the physics of light to pull information out of an object of interest—a group of cells, for example. X-rays and MRIs can detect tumors in the body. Proton therapy and radiation help treat cancer in millions of patients. Recently, however, some physicists have asserted that they can do more for cancer than just develop fancy instruments. Perhaps, they suggest, cancer has resisted a decades-long assault that has cost more than $100 billion partly because the assault has been missing the conceptual artillery of physics.

Image caption: Andrew Ewald
In 2009, a group of physicists convinced the National Cancer Institute, the country's top funder of cancer research, to launch a network of "physical science oncology centers" dedicated to this idea. Physicists from fields as distant from cell biology as cosmology were set loose with millions of dollars to pursue ideas that most biologists weren't interested in—that cancer could be a reversion to a primitive evolutionary program, for example, or that game theory and network theory could help develop a fundamental understanding of the disease. It was a risky idea, says Larry Nagahara, who managed the program for several years before joining the Whiting School of Engineering, and what fruits it ultimately bears remain to be seen.
Ewald—43, bespectacled and clean-shaven with a well-managed mop of sandy blonde hair—isn't one of those physicists who parachuted into cancer biology after establishing a career doing something else. His migration to biology began while he was majoring in physics at Haverford College. A genetics course and a stint in a cancer research center turned him on to the richness and complexity of biology. He went to Caltech for graduate school, thinking he would study the physics of cells and other biological structures—"the most obvious overlap between physics and biology," he says—but he was seduced by a time-lapse movie of the brain in formation that his adviser showed him. As a postdoctoral researcher at UCSF, he had had another pivotal experience when he heard his adviser describe how lungs develop in an embryo. He threw himself into studying how cells form complex tissues, organs, and—when the process goes awry—tumors.
When Ewald reached Johns Hopkins as a new assistant professor in 2008, he knew the grim statistics of cancer morbidity and mortality. Then, two months after he started his position, his father was diagnosed with metastatic colon cancer. "Metastatic" made this a devastating diagnosis: Metastasis is responsible for nine in 10 cancer deaths. Surgeons can often cut out a primary tumor, but there's no way to find and excise dozens or hundreds of far-flung growths, many of which don't show up on scans. Ewald's father died less than four months after his cancer was discovered. It was particularly frustrating, Ewald says, that even Johns Hopkins' world-class doctors did not understand cancer well enough to help. "My only consolation was that I had already organized my research and lab around closing this gap."
Biologists' picture of metastasis, it turned out, was particularly murky. As far back as the 1950s, scientists were finding evidence that both single cancer cells and cell clusters could circulate in the blood. Moreover, patients with clusters in their bloodstream fared worse than those without them. Yet researchers largely ignored the clusters and came to think of metastasis as a process of single cells breaking away from tumors, like lone wolves striking out from their packs for new territory. Ewald suspected that biologists had gone too far, in an Einsteinian sense. "You want to study model systems for cancer that are as simple as possible but not simpler," he says. "You want to have the key features of the system still represented."
Those key features include not just the cells' migration but their specific environment. Cancer cells are constantly pushing and pulling on collagen, a tough yet flexible fibrous protein that fills much of the space outside cells; the density and orientation of collagen can also affect nearby cells. As a postdoc, Ewald had developed a technique to embed cells inside gels designed to mimic collagen. He'd done so to study how organs develop; the same method, he found, allowed him to create lifelike models of cancer cells' environment. But it also posed a challenge: Microscopes are often designed to take images of a single spot on a simple 2-D slide or dish. That's where Ewald brought to bear a physicist's technical tool set. With his physics training, he understood how light is absorbed and reflected and how it can be manipulated to yield more information. He reconfigured his microscopes to image hundreds of positions in quick succession while staying in perfect focus. And he replaced the light filters. Microscope designers typically enhance their instruments' resolution by blasting a sample with as much light as possible. But light that bright kills cells. "Bringing in more light looks fantastic for a few images," Ewald says. "Then the sample dies." He took a different tack: He carefully guided the scope's light beams to make each photon do more work. He managed to supercharge his resolution while keeping light levels low enough for cells to survive indefinitely. He wrote software that made the microscope take a picture every 10 to 20 minutes for up to 100 hours. He calls the technique 4-D confocal microscopy—the fourth dimension being time. He calibrated his equipment to collect not just images but numbers—quantitative measures of cells' positions, velocities, and trajectories.
All scientists measure, but that can mean different things. One might study metastasis by implanting a tumor in a mouse, then observing whether the tumor spreads. If it does, that's a 1; if not, it's a 0. Repeat and build a dataset that tells you something about the relative tendency of certain tumors to spread. But that does not measure how the malignant cells move from one site to another. Ewald's movies provide immensely more data—they reveal the precise physics of cancer's motion. "Andy's work is very quantitative—he measures everything," says Denis Wirtz, a professor of chemical and biomolecular engineering at Johns Hopkins who studies the physical forces on cancer cells. "He knows his biology, his physics, and his optics." This rare combination of expertise in a single person has been key to his success, Wirtz says.

For their breast cancer study, Ewald and his team collected tumors from various patients. No two tumors were alike, reflecting the complex reality of the disease, plus each had a throng of cells with different genes activated, expressing different proteins. The researchers chopped the tumors into roughly spherical blobs of 200 to 1,000 cells each—known as "organoids"—and embedded them in gels; each organoid represented the full cellular diversity of the original tumor. The gels were made of various materials, enabling the researchers to see how the cancer cells behave in different environments they might encounter in the body. "Because we can generate 200,000 to 300,000 of these organoids per tumor, we can put them in as many microenvironments as we can dream up," Ewald says.
To tell what individual cells in the tumors were doing, the researchers labeled them with fluorescent markers. Over hours, many of the clumps grew those long, thin tentacles that reached into the surrounding gel. Ewald and his team found that nearly all the cells at the ends of the tentacles—the ones leading the charge—were labeled green. That meant they contained keratin 14, or K14, a protein that helps give cells their structure and connects them to each other. The follower cells didn't have K14.
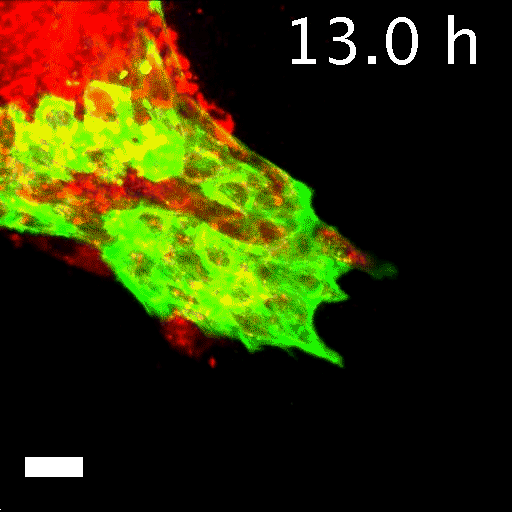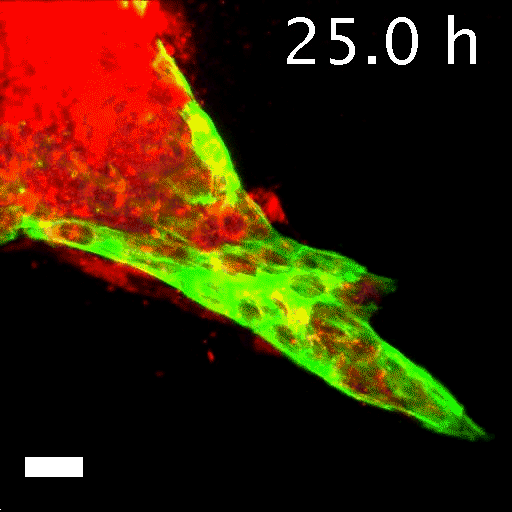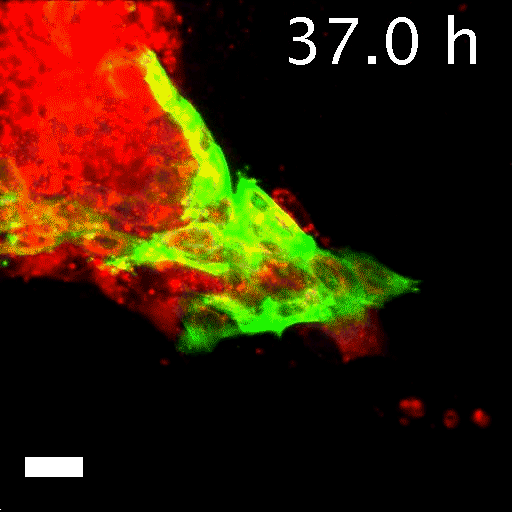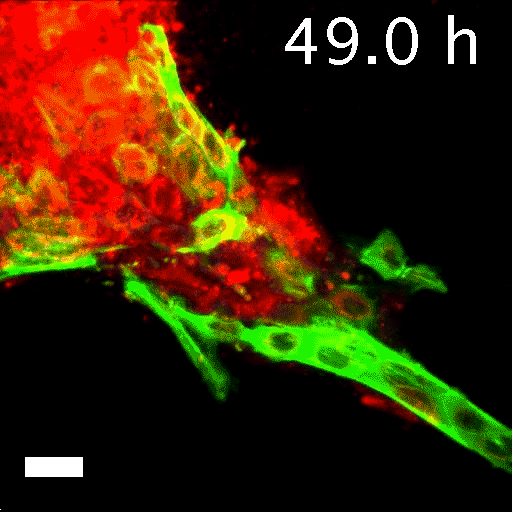
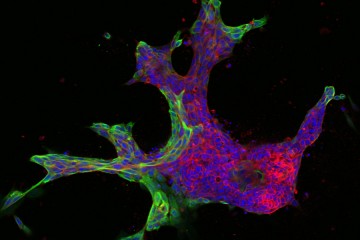
Image caption: Invasive leader cells, dyed green, push into surrounding tissues and carry less invasive cancer cells in red along for the ride. Movie originally published in Cheung et al, Cell.
Image credit: Courtesy of Andrew Ewald
This was a surprise. K14 indicates a relatively rare type of cell, known as an epithelial cell, that lines the outsides of many organs and tumors. According to the standard paradigm, epithelial cells are too sticky and sedentary to separate from tumors and move through the body. Indeed, the researchers had labeled epithelial cells to differentiate them from the cells they thought would metastasize. Now it seemed that the epithelial cells were not just involved in metastasis, they were essential to it.
But Ewald was wary about reading too much into the initial result. "Everyone was saying that cancer is not one disease but many diseases," he recalls. "So it was reasonable [for him and his colleagues] to think that K14 would only explain a subset of cancers." When the team tested the two other main types of breast cancer, both sent out cell groups with K14-expressing leaders. Moreover, when the researchers applied a common technique to stop the cells from making K14, the tumors no longer invaded the gel.
Multicell groups could invade the collagen matrix—that seemed clear. And those groups were invariably led by cells that seemed specialized for the task. But did that prove that malignant cell clusters can actually make it through the bloodstream to seed tumors elsewhere in the body? Hardly. Moving into the environment next door is only the first of many hurdles a breast cancer cell must overcome to successfully colonize, say, a lung, which from the cell's point of view is another country. "Invasion has been seen almost as synonymous with metastasis," Ewald says, but he points out that the phenomenon is far more complex than simply poking into nearby collagen. "Cells have to invade, but they also need to get into the blood vessels, they need to survive in the blood and escape the immune cells, and then land in distant organs, survive there, and eventually grow out." If his cells couldn't complete that progression, his finding might be an interesting curiosity but far short of suggesting a new paradigm of how cancer actually kills people.
So Ewald and his colleagues next implanted tumors in mice. The researchers labeled some of the tumor cells green and some red. (In this experiment the labels did not correspond to specific cell types.) Ewald's team took blood samples from the mice and found multicolored cell clusters made mostly of K14-positive cells. Six to eight weeks after implanting the cells, the scientists found new growths with both green and red cells in the mice's lungs, and now the K14-negative cells were taking over again. Clearly, cell clumps had traveled as a unit from the breast to the lung. The researchers determined that at least 97 percent of the new tumors arose from such clusters; almost none were seeded by single cells. That meant, at least for these breast tumors in these mice, that individual cells traveling solo apparently did little to spread cancer throughout the body.
In Ewald's studies, K14 shows up again and again in leader cells. When it's not there, leader cells don't lead. Perhaps, Ewald speculates, grouping actually helps cancer cells complete metastasis. Individual cells, like individual people, do some things better and other things worse, depending on the genes and molecules that are active in them. The cells out in front may be great at pushing through collagen and into the bloodstream, for example, but they might be comparatively poor colonizers. Building new tumors after settling into a new organ is where the follower cells might show their strength. The "terrifying implication," Ewald says, is that these multiclonal metastases will likely be much harder to kill because doctors probably won't be able to target them with just one drug.
He thinks his finding could have a major upside: It could help predict when a cancer is likely to metastasize. Pathologists diagnose cancer mainly using a century-old visual inspection system known as staging; the procedure notes the size of a tumor, whether it has spread into nearby lymph nodes, and whether metastases can be found. They have no good way to predict which tumors will metastasize, so they often treat all tumors aggressively on the chance they'll spread if not killed. That results in some patients suffering through unnecessary chemotherapy, and, some scientists suspect, providing drug-resistant cancer cells a pathway to more easily take over after chemo wipes out the drug-susceptible ones. Ewald's findings could make pathology more powerful and precise. For example, K14 could be a potent marker that tells pathologists when to go scorched-earth on a tumor and when to hold the artillery.
But there is more work to do. Ewald is now working with clinicians to replicate his findings in a wider range of breast tumor samples, to prove that K14 is universally relevant. And he needs to show that K14 detection actually provides useful information to pathologists and clinicians beyond what's available through existing tests. He's also working with pathologists to test whether K14 can predict recurrence in patients after their primary tumors are removed. (It does in mice, but results in mice often do not translate to humans.) "We're actually optimistic we'll be able to develop a good test of which patients will metastasize," he says.

By the time Ewald's team published results in early 2016 in the Proceedings of the National Academy of Sciences, two other research groups had also reported multiclonal metastases—one using breast cancer cells, the other using pancreatic cancer cells. Ewald and his postdoc Kevin Cheung (now at the Fred Hutchinson Cancer Research Center in Seattle) published a follow-up paper in Science summarizing the trio of results. "Although conventional models suggest that metastases are seeded by single cells from the primary tumor, there is growing evidence that seeding requires the collective action of tumor cells traveling together in clusters," the two wrote.
Other researchers besides Ewald had observed groups of cells leaving a tumor. Few, if any, had tracked the groups all the way to a new organ. "Andy's work is among the first, if not the first, to really show that [tumors] don't need to individualize cells to undergo full metastasis," says Peter Friedl, a biologist at Radboud University in the Netherlands, whose movies of cancer cells in motion helped convince cancer biologists that collective invasion is important. He notes, however, that it's not clear whether all cancers can spread in this multicellular way or whether it's a strategy that only some cancers employ, because so far Ewald and others have proved that collective metastasis occurs in only a few types.
Meanwhile, Ewald has launched a new project that would have been impossible even 10 years ago when he started at Johns Hopkins. His movies had revealed that metastasizing tumors often assume specific shapes—multi-tentacled tumors resemble starfish, for example. He showed some of the tumor images to Joel Bader in Johns Hopkins' Department of Biomedical Engineering. Bader recognized that the shapes were not just pretty; they could also be used as inputs for machine learning, the powerful computational method that is revolutionizing human endeavors from image analysis to chess to how we search the internet. Ewald and Bader have just received a grant to use computers to analyze hundreds of thousands of organoid shapes, rank them according to how invasive they are, and attempt to correlate the invasiveness with genes and proteins that are active during metastasis. The researchers hope this will yield a catalog of new potential drug targets. In a few years, if the project succeeds, machine learning may start to revolutionize biologists' understanding of what really drives metastasis. "We have 1,000-page textbooks on tumor shapes, but it's still not clear what's important," Ewald says. Again—searching for the fundamental simplicity.
On a recent morning, Veena Padmanaban, a graduate student in Ewald's lab, sat in a darkened room. Refrigerators nearby gave off a persistent background drone. Padmanaban was looking at microscope images of organoids made from mouse breast tumors. "We saw that most metastases in mice are caused by cell clusters," Padmanaban says. "What happens if we play around with that?" She had created tumors with and without E-cadherin, a protein that cells use to hold on to each other, like kindergartners holding hands while walking to the playground. In a popular view of metastasis that has long held sway over the field, for a cell to leave a tumor, it must stop producing E-cadherin and set off alone, like a child taking off down the sidewalk. But Ewald's team had found in their previous studies on collective invasion that both leader and follower cells continue to produce E-cadherin and use it to stay connected as they make their way into the collagen matrix.
Padmanaban wanted to know whether tumors with E-cadherin are actually more dangerous. She used a joystick to navigate the image field, as in an old Atari video game. Tumors in which she had inactivated the gene that produces the protein did spit out cells, following the conventional paradigm of metastasis. But the mice with these tumors did not end up with metastatic cancer. Apparently, single cells were ready and able to pick up and move but incapable of settling down again, or they got swept up by the immune systems of the mice. Tumors that produce E-cadherin, on the other hand, released fewer total cells into the surrounding matrix, but mice implanted with those tumors soon had lungs peppered with new growths.
Once again, in studying the full complexity, an important underlying simplicity was emerging. Or, perhaps a better way to think of it—that simplicity had been there all along, waiting for the right tools, the right methodology, and the right minds to reveal it.
Posted in Health, Science+Technology
Tagged cancer, physics, cell biology, cancer metastasis